
Calculating Hydronium ion concentration from pH (and identifying molecules based on pH) : r/chemhelp
![Definition pH and pOH. Given pH, pOH, [H 3 O + ] or [OH¯], calculate the remaining values. Calculate Ka/Kb, given the pH or pOH and the concentration. - ppt download Definition pH and pOH. Given pH, pOH, [H 3 O + ] or [OH¯], calculate the remaining values. Calculate Ka/Kb, given the pH or pOH and the concentration. - ppt download](https://images.slideplayer.com/34/8310807/slides/slide_10.jpg)
Definition pH and pOH. Given pH, pOH, [H 3 O + ] or [OH¯], calculate the remaining values. Calculate Ka/Kb, given the pH or pOH and the concentration. - ppt download
![SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice, SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice,](https://cdn.numerade.com/previews/c92ef7f1-c83f-4027-9f05-6b11ebe1b21e_large.jpg)
SOLVED:MATHEMATICAL Calculate the hydrogen ion concentration, [H^+] for each of the following materials: (a) Saliva, pH 6.5 (b) Intracellular fluid of liver, pH 6.9 (c) Tomato juice, pH 4.3 (d) Grapefruit juice,
What is the concentration of hydrogen ion and hydroxide ions of solution which has pH of 4.87? - Quora

SOLVED:Calculate the hydrogen ion concentration in mol/L for each of the following solutions: (a) a solution whose pH is 5.20, (b) a solution whose pH is 16.00,(c) a solution whose hydroxide concentration
![Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download](https://images.slideplayer.com/17/5302527/slides/slide_3.jpg)
Calculating pH and pOH. pH pH = - log [H + ] [H + ] = the hydrogen ion concentration pH: “potential of hydrogen” - A way of expressing the hydrogen ion. - ppt download










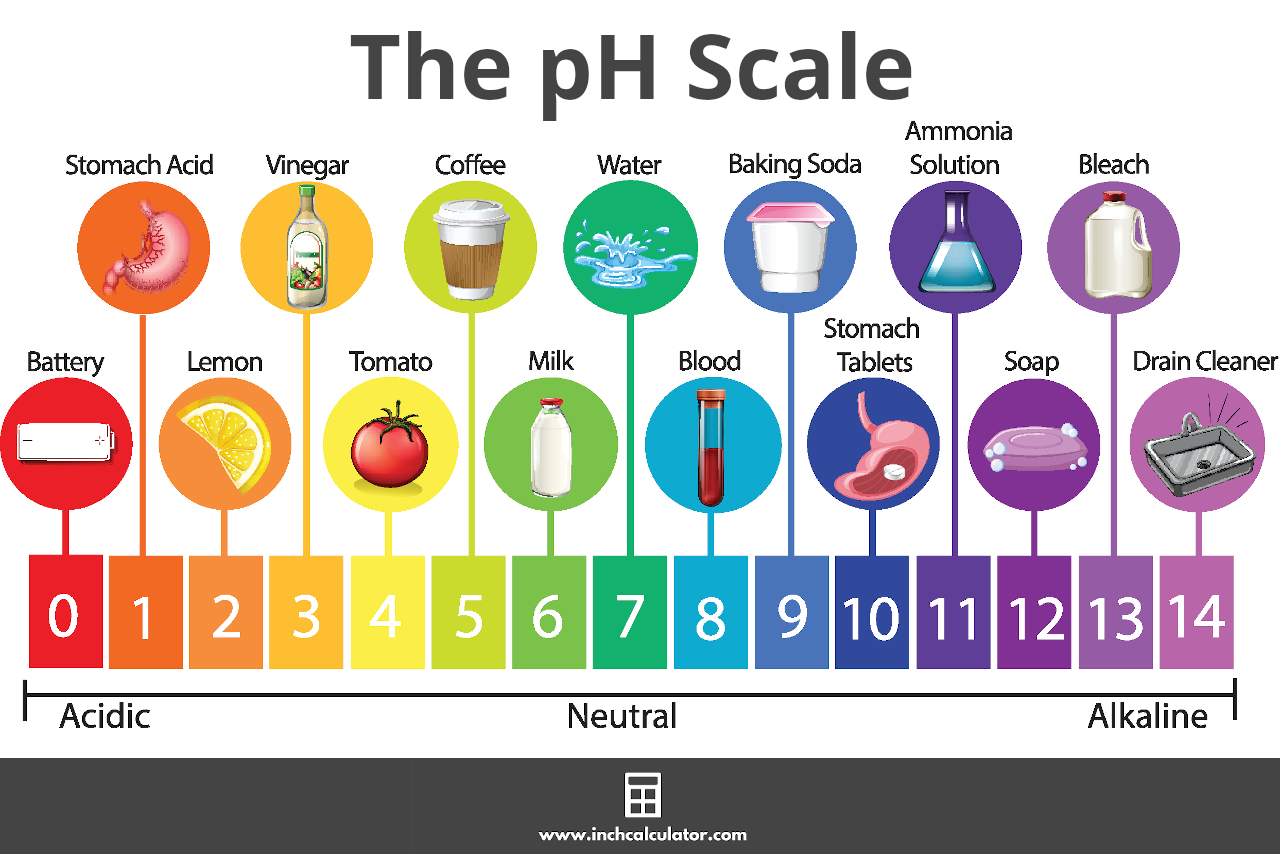
:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)



![Calculating [H+] from pH, Acids & Bases Tutorial - YouTube Calculating [H+] from pH, Acids & Bases Tutorial - YouTube](https://i.ytimg.com/vi/bP-evPgNNUg/maxresdefault.jpg)


